Up until 2024, GLP-1 receptor agonism for body composition meant injecting a peptide weekly. The shift now in motion is two-track: an oral peptide that slips past the gut at deliberately reduced bioavailability (Rybelsus), and a small-molecule GLP-1 RA with conventional pharmaceutical-pill kinetics (Orforglipron, in late-stage Lilly trials). For the audience this site is written for, both reset who the “injectable GLP-1” user actually is - and who it isn’t.
The two tracks
Rybelsus - oral semaglutide that’s still a peptide
- What it is. The same semaglutide molecule as Ozempic and Wegovy, formulated with sodium N-(8-[2-hydroxybenzoyl]amino)caprylate (SNAC), an absorption enhancer that briefly opens a path for a peptide to cross the gastric mucosa. Approved by the FDA in 2019 for type 2 diabetes (3, 7, 14 mg daily); the higher-dose 25 and 50 mg formulations (PIONEER PLUS / OASIS-1) for obesity have moved through trials more recently.
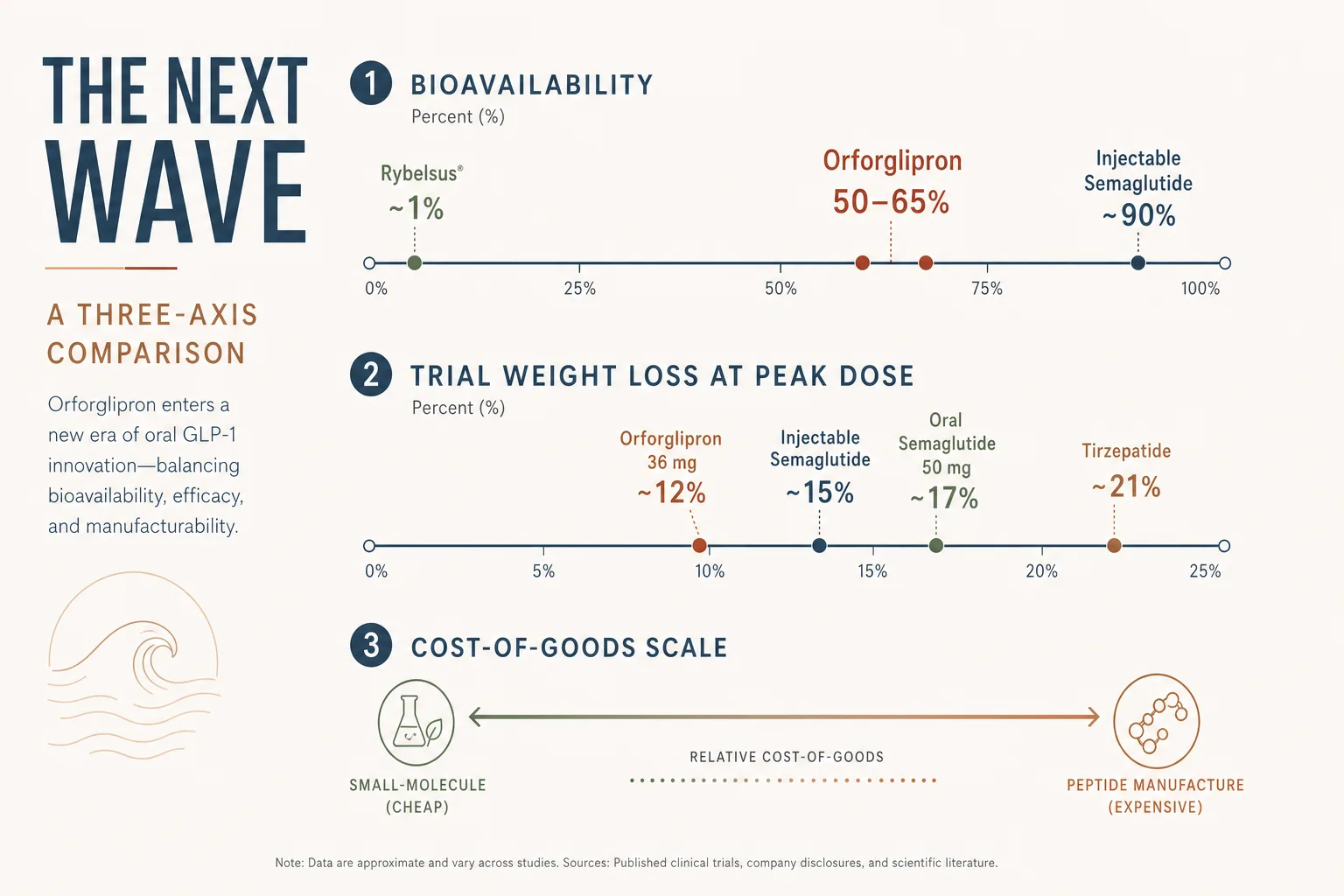
- Bioavailability. Roughly 1% - meaning a 14 mg oral tablet is in the same systemic-exposure neighbourhood as ~140 mcg of subcutaneous semaglutide. The dosage gap reflects how much is wasted by gut degradation. The practical implication is fasting requirements: Rybelsus must be taken on an empty stomach, with at most 4 oz of water, 30 minutes before any food, drink, or other oral medication. Failure here cuts absorption to near-zero.
- Trial efficacy. PIONEER 4 (T2DM, 14 mg vs liraglutide 1.8 mg SC) showed comparable HbA1c reduction. OASIS-1 (obesity, 50 mg daily) showed 17.4% mean weight loss at 68 weeks vs 1.8% on placebo - in the neighbourhood of injectable semaglutide 2.4 mg weekly (~14.9% in STEP 1).
- Where it fits. Users who can keep the fasting-window discipline and hate weekly injections. Travel-friendly. Slightly more flexible miss-a-day tolerance than the weekly injection (one missed daily pill is a smaller deviation than one missed weekly shot relative to total exposure).
- Where it doesn’t fit. Users who are inconsistent about morning fasting windows. The fasting requirement is not a suggestion - eat or drink coffee within 30 minutes of the pill and absorption craters.
Orforglipron - small molecule, ordinary pill kinetics
- What it is. A non-peptide, orally bioavailable GLP-1 receptor agonist developed by Eli Lilly. Daily dosing, no fasting requirement, no peptide-stability constraints - pharmacologically behaves like a conventional small-molecule pharmaceutical.
- Why this matters. Manufacturing a small molecule is orders of magnitude cheaper than recombinant peptide production. If Orforglipron approves and the price reflects manufacturing cost, the GLP-1 access ceiling shifts dramatically - including in territories where GLP-1 supply has been a chronic limiter.
- Trial efficacy. ATTAIN-1 (obesity, phase 3) and ACHIEVE-1 (T2DM, phase 3) reported in 2025 with 36 mg daily showing weight loss in the 11–15% range at 72 weeks - not at tirzepatide’s level (~21% on 15 mg in SURMOUNT-1) but competitive with injectable semaglutide and notable for an oral daily pill.
- Regulatory status. NDA submission was expected in late 2025 with approval on the 2026 timeline if filing tracked cleanly; verify current status against the FDA tracker before assuming availability through approved channels. Compounded / grey-market versions are biochemically harder to verify because the compound is a small molecule, not a peptide - HPLC-MS still works but the standards used by community reference labs are mostly peptide-tuned.
- Where it fits. Future default first-line for users who don’t need tirzepatide-tier efficacy. Lower side-effect burden trends in the trial data than injectable semaglutide - GI events still common but more dose-titratable.
What this changes for the catalogue user
- The injection question becomes optional. For weight-loss-only goals at moderate efficacy targets, an oral pill will almost always be the friction-minimising choice. Injectable GLP-1 stays the answer for users wanting tirzepatide’s higher ceiling, retatrutide’s triple agonism, or weekly-cadence convenience.
- The supply / cost story shifts. Compounded GLP-1 popularity is downstream of supply constraints and pharmacy pricing. If Orforglipron arrives at small-molecule pricing through approved channels, the compounded grey-market case weakens for the user population it currently serves.
- The verification problem inverts. Injectable grey-market semaglutide / tirzepatide can be HPLC-verified by community labs against peptide standards. Small-molecule Orforglipron isn’t a peptide; the verification toolchain that operators have built doesn’t cleanly apply.
- Muscle preservation principles still apply. Any GLP-1 RA at body-comp-altering exposure raises the same protein-and-resistance-training question covered in GLP-1 and Muscle Preservation. Oral or injected, the deficit does the lean-mass damage if discipline isn’t there.
Decision frame for the next 18–24 months
- Currently on injectable semaglutide and well-tolerating?
→ No need to switch. The data quality on injectable is the deepest; the cardiovascular MACE benefit (SELECT 2023) is on injectable specifically. - Considering starting GLP-1 right now, modest weight-loss
goal?
→ Rybelsus 25 or 50 mg is a reasonable starting point if morning-fasting discipline is realistic. If not, injectable semaglutide via approved channel. - Considering starting and high weight-loss goal?
→ Tirzepatide first-line. Oral options haven’t hit tirzepatide’s 21% trial number. - Wait for Orforglipron?
→ Reasonable for users with mild-to-moderate goals and no urgency. The approval timing and post-launch pricing are the open questions; FDA filing has been progressing. - Tested athlete?
→ All current GLP-1 receptor agonists, including Orforglipron and oral semaglutide, sit in the same WADA category as the injectables. The route doesn’t change category. See WADA Testing and Detection.
What stops people
- Treating Rybelsus as a casual oral. The fasting window is the protocol - eat or drink coffee within 30 min and the dose drops to near-zero. Users who can’t hold that discipline get unreliable response and decide GLP-1 doesn’t work for them.
- Comparing peak doses across compounds without context. Rybelsus 50 mg ≈ injectable semaglutide 2.4 mg in systemic exposure terms. The dose number isn’t comparable on its own.
- Buying compounded oral semaglutide for the convenience. If the formulation doesn’t include the SNAC absorption enhancer (or a verified equivalent), the bioavailability story falls apart and you’re absorbing nothing useful.
- Assuming “oral” means “safer.” The same nausea, GI events, and titration discipline apply to all routes of GLP-1 RA. Oral isn’t a softer experience - it’s a different shape of the same physiology.
Cross-references
- Semaglutide vs Tirzepatide vs Retatrutide - the per-compound choice across the injectable family.
- GLP-1 and Muscle Preservation - the discipline that determines body-comp outcome regardless of route.
- GLP-1 Titration Schedule - for the injectable step-up; oral schedules differ but the titrate-and-hold framing is the same.
- Fat Loss application - the broader decision tree across GLP-1 / GH-axis / HGH fragment families.