This article is for tested athletes - collegiate, USADA-pool, Olympic-stream, league-tested (UFC, MLB, NFL, NHL, FIFA), and any competitive sanction body that uses the WADA list. If you are not in that population, this is not a tactical evasion guide; it’s a constraint map. Almost every peptide on this site falls into a banned category. Knowing which ones, what testing actually looks like, and what “detection window” numbers really mean is the difference between an informed protocol decision and a sanction.
The WADA Prohibited List is updated annually (current edition published each January). Categories shift slightly year to year; the broad shape below is stable but verify against the current published list before any competition-window decision.
How the catalogue maps to WADA categories
S0 - Non-Approved Substances
Anything pharmacologically active that’s not approved for human therapeutic use by any government health authority worldwide. Catch-all for the research- chemical end of the catalogue. Banned at all times (in and out of competition).
- BPC-157 - explicitly named in WADA guidance.
- AOD-9604 - never approved for obesity (the development program failed); falls under S0.
- Most novel research peptides without a regulatory pathway end up here by default.
S2 - Peptide Hormones, Growth Factors, Related Substances and Mimetics
The biggest bucket for this catalogue. Banned at all times. Sub-categories matter because testing methodology differs.
- S2.1 - Erythropoietin family. Not really a peptide- catalogue concern but listed for completeness.
- S2.2 - Peptide hormones and their releasing factors. Includes Somatropin (rHGH), Mod GRF, CJC-1295, sermorelin, Tesamorelin, all GH-releasing peptides (Ipamorelin and friends).
- S2.3 - Growth factors. IGF-1 LR3, IGF-1-DES, PEG-MGF, mecasermin, fibroblast/hepatocyte/ vascular-endothelial growth factors. The whole growth-factor portion of the catalogue.
- S2 GLP-1 question. The GLP-1 family (semaglutide, tirzepatide, retatrutide, liraglutide) sits in a more nuanced position. They’re peptide hormones, but WADA has not blanket- banned GLP-1 receptor agonists for performance reasons. As of the current list, they’re not on the prohibited list for tested competition per se, though weight-class sports can still raise issues if a TUE isn’t in place. Verify against the current list because this category has been moving.
- Melanotan II and PT-141 sit in a grey zone - not explicitly listed by name on most years, but the broad S2 hormone-mimetic language and the S0 catch-all both apply to MT-II. Functionally treat as banned.
S5 - Diuretics and Masking Agents
Not directly relevant to the peptide catalogue, but worth flagging: stacks that combine peptides with diuretics for water-cut purposes hit two categories at once.
P1 / P2 - In-competition only categories
Beta-blockers and other in-competition-only categories don’t intersect with the catalogue meaningfully. Out-of-competition testing covers the S0/S2 hits regardless.
How detection actually works (and where it doesn’t)
“Detection window” numbers from forums are mostly fiction. They conflate plasma half-life with assay sensitivity with sample-collection cadence. The current testing methodology has three real pathways:
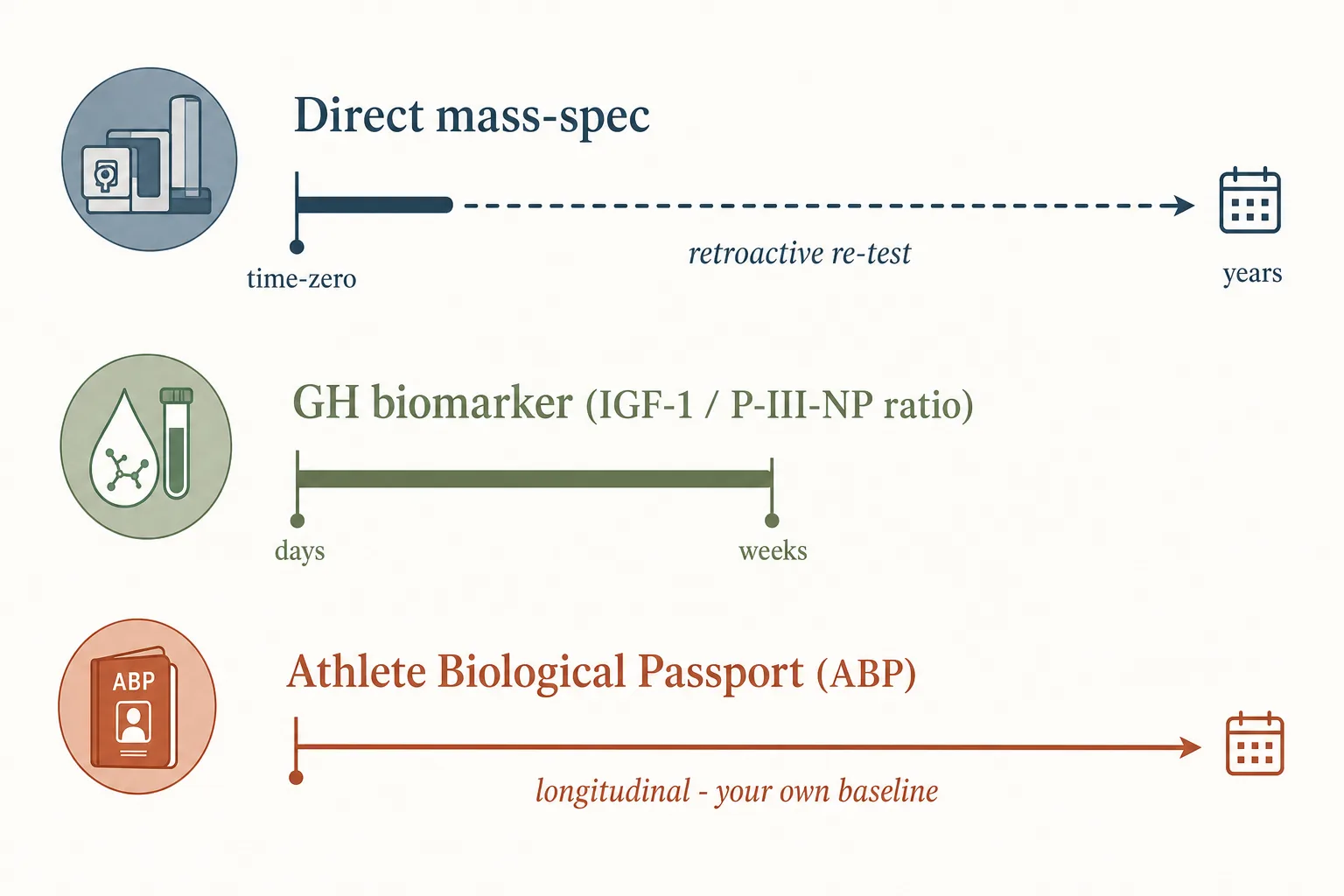
1. Direct mass-spec identification
- Some peptides have validated LC-MS/MS or LC-HRMS assays that detect the parent compound or a known metabolite. GHRPs and GHRH analogues are routinely identifiable on current WADA lab assays at sub-ng/mL levels in urine.
- Detection-window question becomes “at what dose, how soon after administration, and through which route” - not a fixed number.
- Worth knowing: WADA labs increasingly use long-term-storage of B-samples and retroactive re-analysis. A sample taken in 2024 and stored can be re-tested in 2030 against an assay that didn’t exist when the sample was collected.
2. Indirect / biomarker detection
- The GH biomarker test. Measures IGF-1 and procollagen-III- N-terminal-peptide (P-III-NP) ratios. This is the standard way exogenous GH gets flagged on Olympic-stream testing - the assay doesn’t need to detect GH itself, just the disturbed downstream signature. Detection windows are days to weeks past last administration depending on protocol.
- The Athlete Biological Passport (ABP) endocrine module. Tracks IGF-1 and GH markers longitudinally for each athlete. Out-of-range excursions trigger investigation even without a positive direct test. Hard to beat with intermittent peptide use because you become your own baseline.
- The GH isoform test. Distinguishes recombinant 22-kDa GH from the natural mix of pituitary isoforms. Short detection window (~24–48 hours post administration in most protocols), so it’s the more easily timed-around assay.
3. Prohibited-substance-mimic detection
- For peptides where direct assays aren’t in routine deployment, the sample is screened by general LC-MS/MS for known peptide sequences. Sequence-based detection works well for compounds with established MS fingerprints (GHRPs, the BPC-157 fragment pattern, IGF-1 LR3’s Arg-Tyr extension) and badly for novel research peptides.
- A negative routine screen does not mean “not detectable” - it means “not on this lab’s targeted panel today.” Retroactive re-analysis applies here too.
What “detection window” actually means
Forum numbers like “Ipamorelin clears in 6 hours” almost always reflect the plasma half-life, not the time the lab can no longer find it. The real number depends on:
- Assay sensitivity. A lab that can detect a metabolite at 100 pg/mL has a different effective window than one stuck at 1 ng/mL. Sensitivity improves year-on-year - what was undetectable in 2018 is routinely caught now.
- Route and dose. Higher doses extend the window, sometimes non-linearly. SC vs IM matters because of release kinetics.
- Sample type. Urine has a longer effective window for many peptide metabolites than blood. The trend is toward dried-blood-spot collection for some assays, which changes the math again.
- Indirect markers. Even if the parent compound is gone, IGF-1 elevation on an ABP curve takes weeks to resolve. That’s the window that actually matters for GH-axis users.
Practical implication: anyone in a tested pool who calculates a detection-window protocol against forum numbers is making a decision against fictional numbers. The honest answer is that for most peptides on this site, the current sensitive assays mean the operational detection window is “weeks to months past last use” for indirect markers, and indeterminate for retroactive re-analysis.
The TUE pathway
A Therapeutic Use Exemption is the only legal route through the prohibited list for a tested athlete. The criteria are strict:
- The substance must be needed to treat a diagnosed medical condition.
- Without it, the athlete’s health would be significantly impaired.
- The substance does not produce additional performance enhancement beyond restoring the athlete’s normal state of health.
- No reasonable alternative non-prohibited treatment exists.
GH-deficient athletes can sometimes obtain TUEs for somatropin replacement. GLP-1 TUEs for diabetes are common and uncontroversial. TUEs for “recovery” or “anti-aging” are not granted; the criteria explicitly exclude performance-restoration framing. Submission goes through the athlete’s national anti-doping organisation (NADO) or international federation, not directly to WADA.
League-specific notes
- USADA pool / Olympic stream. Strictest. Out-of-competition testing without notice. ABP is fully deployed. Retroactive re-analysis on long-term storage. Treat all S0/S2 substances as completely off the table without a TUE.
- UFC (USADA partnership ended late 2023, replaced by Combat Sports Anti-Doping in 2024). Still WADA-aligned but the testing partner differs. Direction has been toward more out-of-competition testing, not less.
- NFL. Has its own list and testing program, generally tracks WADA on peptides but with looser out-of-competition coverage. GH testing is deployed; biomarker test in use.
- MLB. Joint Drug Agreement aligned with WADA for peptide hormones. GH biomarker test added in 2014.
- NCAA. Prohibits the WADA list. Testing coverage varies enormously by school, sport and conference. Don’t mistake light testing for permission.
- FIFA / UEFA football. WADA-aligned. ABP module deployed.
- Powerlifting / strongman federations. Vary widely. “Tested” federations align with WADA; “untested” ones explicitly do not test.
What stops people
- Trusting forum detection-window numbers. Most are wrong by an order of magnitude on indirect markers, and don’t account for retroactive testing at all.
- Assuming “not on the list by name” means legal. Both S0 (non-approved substances) and S2’s “and related substances and mimetics” language are catch-alls. A novel research peptide that’s not specifically named is still functionally banned.
- Microdosing as a detection-evasion strategy. Reduces direct- detection window but doesn’t reduce ABP signal much. The IGF-1 elevation from sustained low-dose protocols is exactly the longitudinal pattern the ABP is designed to catch.
- Forgetting about long-term sample storage. WADA stores B- samples for up to 10 years. A clean test today is not a permanent clean test if the assay improves.
- Assuming TUE is a back-door. The criteria are reviewed by physicians familiar with the substance. “Recovery” or “wellness” framing fails. Legitimate medical conditions with documentation succeed; nothing else does.
- Buying counterfeit and getting flagged anyway. A contaminated product can produce a positive for something the user didn’t knowingly take. The strict-liability framework means the athlete bears the consequences regardless. See Sourcing and Verification.
Cross-references
- Sourcing and Verification - counterfeit risk, which intersects with WADA strict-liability framework.
- Cycling Strategies - for the cycle-pattern decisions that affect detection-window math.
- Bloodwork for Peptide Users - IGF-1 monitoring is also a passive indicator of what an ABP module would see.
- GH Axis Playbook - the GH-axis decisions where the WADA constraint matters most.
- Peptides for Endurance Athletes - the population that runs into the WADA constraint most consistently; in-season vs off-season cycling decisions sit on top of this article's framework.