Three compounds dominate the modern incretin space: Semaglutide (Ozempic, Wegovy, Rybelsus), Tirzepatide (Mounjaro, Zepbound), and Retatrutide (investigational, LY3437943). They are often discussed as a single category - “the GLP-1s” - but they target different combinations of receptors, hit different efficacy ceilings, carry different side-effect loads, and sit at different points on the regulatory timeline. This article walks through each one, then ends in a decision tree for picking between them.
The receptor stack
Three gut/pancreatic hormone receptors do almost all of the work here:
- GLP-1 receptor - central appetite suppression, glucose-dependent insulin secretion, slowed gastric emptying. The original target. This is what makes “food noise” quiet down.
- GIP receptor - incretin signaling that synergizes with GLP-1 on insulin sensitivity and appears to soften the GI side-effect curve at matched efficacy. Mechanism is still partially debated, but the trial outcomes are clear.
- Glucagon receptor - activation here raises energy expenditure and drives fat oxidation, at the cost of higher resting heart rate and a more “activated” feel. This is what separates retatrutide from the others.
Semaglutide hits one of those (GLP-1). Tirzepatide hits two (GLP-1 + GIP). Retatrutide hits all three. The efficacy data tracks that progression: more receptors engaged, more weight loss, more side-effect surface area to manage.

Semaglutide
- Class: Selective GLP-1 receptor agonist.
- Half-life: ~7 days. Once-weekly injection (Ozempic, Wegovy) or daily oral tablet (Rybelsus, much lower bioavailability).
- Standard dose range: 0.25 mg titrated up to 2.4 mg weekly for weight loss; 0.5–1.0 mg for diabetes. Step up every four weeks.
- Trial efficacy: STEP 1 (Wilding 2021, NEJM) showed 14.9% mean weight loss at 68 weeks vs 2.4% on placebo. SELECT (Lincoff 2023, NEJM) showed a 20% reduction in major adverse cardiovascular events in overweight/obese adults without diabetes - a hard outcome, not just a surrogate.
- Side-effect profile: Nausea, vomiting, constipation, occasional gallbladder events. Strongly dose- and titration-dependent. Most users settle once they plateau on a maintenance dose.
- Cost / supply: Approved and widely available, though brand-name pricing is steep without insurance. Compounded grey-market supply has tightened substantially as Novo Nordisk’s shortage status has moved.
- Who it’s for: The default first-line GLP-1 for someone who wants a well-characterized, FDA-approved option with cardiovascular outcome data behind it. Lower weight-loss ceiling than tirzepatide, but the longest track record and the cleanest evidence base.
Tirzepatide
- Class: Dual GIP / GLP-1 receptor agonist (“twincretin”).
- Half-life: ~5 days. Once-weekly subcutaneous.
- Standard dose range: 2.5 mg titrated up to 15 mg weekly. Step up every four weeks. Many users hold at 5 mg or 10 mg as long as weight loss continues; bigger isn’t always better here, and the side-effect curve climbs faster than the efficacy curve at the top doses.
- Trial efficacy: SURMOUNT-1 (Jastreboff 2022, NEJM) showed 20.9% mean weight loss at 72 weeks on 15 mg vs 3.1% placebo. SURPASS-2 (head-to-head vs semaglutide 1 mg in type 2 diabetes) showed superiority on both A1C and weight loss across all three tirzepatide arms.
- Side-effect profile: Same GI pattern as semaglutide - nausea, constipation, vomiting if titrated too fast - but several head-to-head signals suggest GI tolerability is incrementally better at matched weight-loss magnitude. Boxed rodent C-cell tumor warning carries over from the GLP-1 class.
- Cost / supply: Approved (Mounjaro for diabetes, Zepbound for obesity). More consistently available than semaglutide through 2025 because it came to market later and had less aggressive demand pile-up. Brand pricing is comparable; insurance coverage is spottier than diabetes indications.
- Who it’s for: The current efficacy ceiling among approved options. The right choice when the goal is maximum weight loss within an FDA-approved framework, or when semaglutide tolerability has been a problem.
Retatrutide
- Class: Triple GLP-1 / GIP / glucagon receptor agonist.
- Half-life: ~6 days. Once-weekly subcutaneous.
- Studied dose range: Phase 2 dose arms ran from 1 mg up to 12 mg once-weekly with stepwise escalation. There is no “standard” outpatient protocol because the drug is investigational.
- Trial efficacy: The phase 2 obesity trial (Jastreboff 2023, NEJM) showed dose-dependent weight loss across all arms over 48 weeks, with the highest-dose arm exceeding the magnitude of any approved incretin therapy. Phase 2a data also support meaningful improvement in liver fat in MASLD/MASH (Respi 2024, Nature Medicine). The phase 3 cardiovascular and kidney outcomes trial (TRIUMPH-Outcomes, NCT06383390) is recruiting; multi-year outcomes do not yet exist.
- Side-effect profile: Same GI pattern as the other two, but additionally a measurable increase in resting heart rate driven by the glucagon-receptor component. This is the meaningful differentiator. Anyone with a baseline cardiovascular concern, or who is sensitive to stimulants, should weight that profile carefully.
- Cost / supply: Investigational. Any non-trial supply is grey-market research-chemical material with no quality assurance, no standardized concentration, and no clinical monitoring. Treat any non-trial use as substantially higher uncertainty than semaglutide or tirzepatide use, even before considering the missing long-term safety data.
- Who it’s for: Right now, mostly trial participants and a small number of users who are willing to accept a meaningful product-quality uncertainty in exchange for the largest weight-loss signal in the class. For everyone else, wait for the phase 3 readouts.
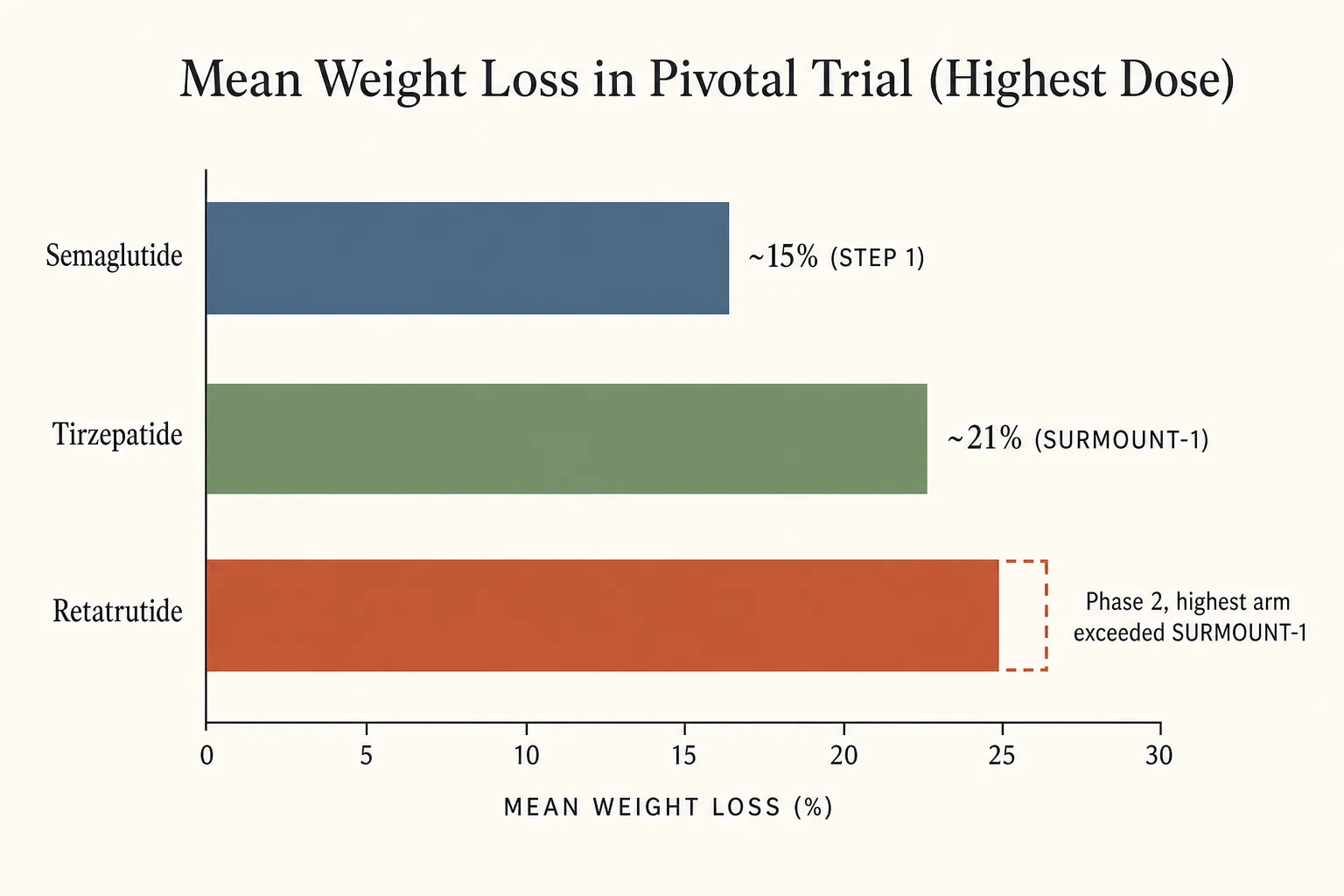
Side-by-side
- Receptor footprint: Semaglutide = GLP-1. Tirzepatide = GLP-1 + GIP. Retatrutide = GLP-1 + GIP + glucagon.
- Mean weight loss in pivotal trial (highest dose): ~15% (STEP 1) < ~21% (SURMOUNT-1) < phase 2 retatrutide (highest arm exceeded SURMOUNT-1 magnitude).
- Cardiovascular outcomes data: Semaglutide has it (SELECT, SUSTAIN-6). Tirzepatide’s SURPASS-CVOT and SURMOUNT-MMO are reading out. Retatrutide’s TRIUMPH-Outcomes is recruiting.
- Heart-rate impact: Modest with semaglutide / tirzepatide. Materially larger with retatrutide because of glucagon-receptor activation.
- Regulatory status: Semaglutide and tirzepatide are FDA-approved. Retatrutide is investigational.
- Supply quality: Approved channels for the first two. Retatrutide is research-chemical only outside trials.
Muscle preservation applies to all three
All incretin therapies drive rapid weight loss in part by cutting appetite hard, and rapid weight loss without effort always strips lean mass alongside fat. The harder the appetite suppression, the more aggressive the protein and resistance-training discipline has to be to compensate. Triple agonism doesn’t change that - it makes the deficit deeper, which is exactly why this matters more, not less, on retatrutide than on semaglutide.
The non-negotiables are the same across the class: protein at roughly 1 g per pound of goal body weight, three or more resistance-training sessions per week with progressive overload, and a slower titration than the package insert allows for if appetite is being crushed too fast to hit those numbers. The per-compound step-up tables and the slow-ramp / split-dosing variants are in the GLP-1 Titration Schedule guide. Bloodwork before and during - HbA1c, fasting glucose, lipids, CBC - closes the loop.
Decision tree
- Want the longest track record, an FDA-approved channel, and confirmed
cardiovascular outcome data?
→ Semaglutide. Accept a lower weight-loss ceiling. - Want the maximum weight loss available within FDA-approved options, and
semaglutide either failed or stalled?
→ Tirzepatide. Better tolerability at matched efficacy and a higher ceiling. Currently the gold standard. - Have already exhausted tirzepatide at top dose for long enough to evaluate, and
accept investigational drug uncertainty?
→ Retatrutide, ideally through a trial. Watch resting heart rate carefully. Source quality is the dominant risk factor outside of trials. - Have a baseline cardiovascular concern (uncontrolled hypertension, arrhythmia
history, beta-blocker dependence)?
→ Stay out of retatrutide territory. Semaglutide or tirzepatide. - Are you a tested athlete (WADA, USADA, league)?
→ All three are subject to scrutiny. Insulins and metabolic modulators are S4 on the WADA list; check current league rules and TUE pathways before any use. This is not a place to assume the rule that applied last season still applies.
What this article doesn’t cover
Liraglutide (Saxenda, Victoza) is omitted - once-daily injection, lower efficacy, and superseded by once-weekly options for almost every use case. Dulaglutide and exenatide similarly. The oral track - Rybelsus and Orforglipron - gets its own writeup in The Oral GLP-1 Era. The pipeline past retatrutide - CagriSema, survodutide, MariTide, the amylin angle - is in CagriSema and the Pipeline Past Retatrutide. PCOS-context use, fertility wash-out timing, and perimenopausal baseline considerations are in Peptides and Female Physiology.