A vendor's certificate of analysis says ">98% by HPLC" and the conversation usually stops there. The number is doing more work than most readers realise - and a lot less than they assume. This is the decoder companion to Sourcing and Verification: that article tells you how to verify; this one tells you what the verification number actually counts, why the same number means different things on a 5-mer versus a 35-mer, and what's hiding in the missing 2% (or 5%, or 10%).
What ">98% main peak" actually measures
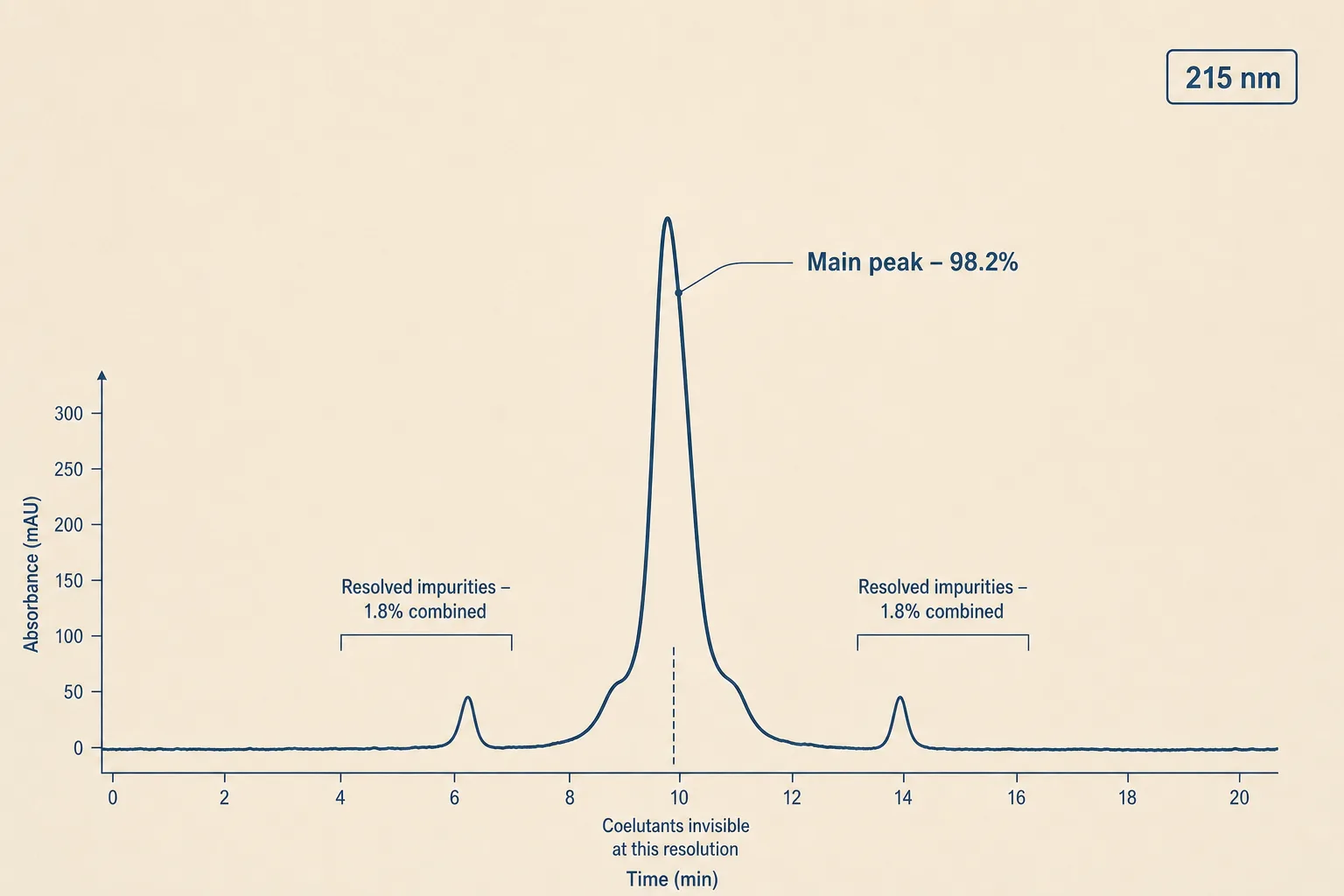
Reverse-phase HPLC separates a peptide sample by hydrophobicity. The detector reports absorbance at 215 nm (or 220, or 280) over time, producing a chromatogram: a baseline with peaks rising above it as components elute. The "main peak percent" is the area of the largest peak divided by the total integrated area of all peaks. 98% main peak means 2% of the absorbance came from something that wasn't the target compound.
- It's a relative number, not an absolute one. The 2% measures peaks the analytical method resolves from the main peak. Coelutants - impurities that come off the column at the same retention time as the target - are invisible to this measurement and roll into the 98%.
- Detection wavelength matters. 215 nm catches peptide bonds; 280 nm catches aromatic residues (Trp, Tyr, Phe). A peptide with no aromatics looks different at 280 than at 215; impurities that absorb mainly elsewhere can be invisible to one wavelength.
- Integration thresholds vary by lab. Tiny peaks near the baseline may or may not be integrated depending on how the lab sets the threshold. Two labs analysing the same vial can report 98.2% and 99.1% from the same chromatogram.
- The number says nothing about identity. A 99% pure sample of the wrong peptide will report 99%. Identity comes from the mass-spec confirmation; purity comes from the HPLC integration. They're different questions on the same COA.
Why long peptides are inherently dirtier
Most catalog peptides come out of solid-phase peptide synthesis (SPPS): a resin-bound first amino acid is extended one residue at a time, with each cycle deprotecting the new N-terminus and coupling the next protected amino acid. Each coupling step has a yield below 100%. Even at 99.5% per-step efficiency - which is excellent in practice - the math compounds:
- 5-mer (4 couplings): 0.9954 ≈ 98.0% theoretical full-length yield.
- 10-mer: 0.9959 ≈ 95.6%.
- 20-mer: 0.99519 ≈ 90.9%.
- 30-mer: 0.99529 ≈ 86.5%.
- 40-mer: 0.99539 ≈ 82.2%.
These are theoretical ceilings before purification. After preparative HPLC purification a vendor can recover a much higher purity from a messy crude - but the cost of that purification climbs sharply with chain length, and the yield drops. A 5-mer hitting 99.5% main peak after a single purification pass is a routine exercise; a 35-mer hitting the same number requires multiple chromatographic steps and produces dramatically less final material per starting batch.
This is why the price-per-milligram curve climbs fast past about 25 residues, and why community testing of long peptides (full-length Thymosin Beta-4 at 43 residues, Follistatin-related fragments, GLP-1 analogues at 30+ residues) routinely shows wider variance than testing of short peptides. The synthesis is harder. The grey-market vendors who hit the same purity numbers as the GMP manufacturers are absorbing real cost to do so; the ones cutting corners are taking the cheap path through one of the failure modes below.
The impurity catalogue: what's actually in the 2%
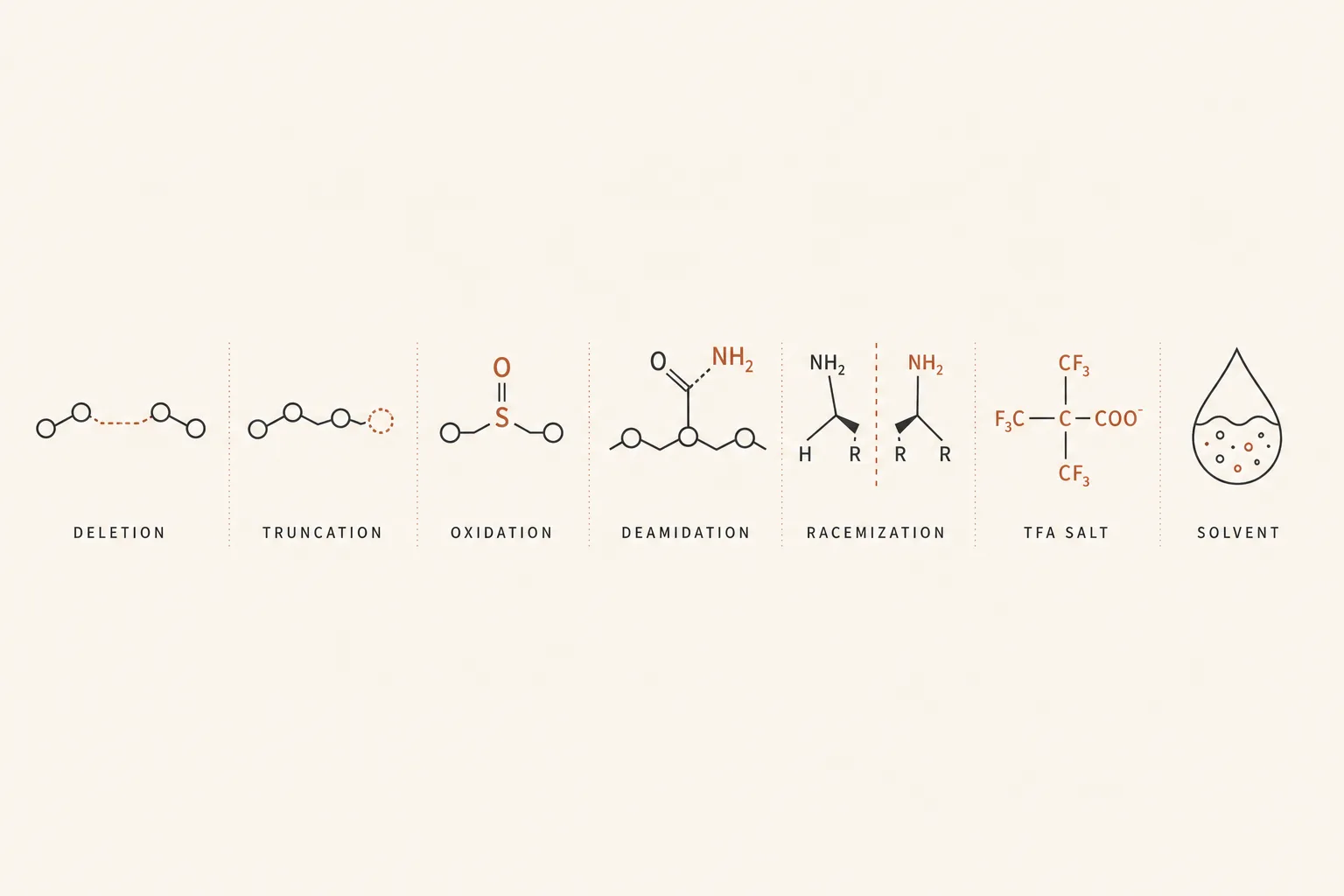
- Deletion sequences. A coupling step that fails produces a peptide missing one residue. Deletion peptides often have similar hydrophobicity to the parent and are the most common "near miss" impurity. A deletion of a non-critical residue may retain partial activity; a deletion in a receptor-contact region is inactive.
- Truncation products. Synthesis that terminates early - usually because of an irreversible side reaction during coupling - produces shorter fragments. These are easier to separate from the parent than deletions but contribute to the overall impurity load on long syntheses.
- Oxidation products. Methionine, cysteine, and tryptophan oxidise on storage and during synthesis. Met → MetO is the most common; the oxidised peptide elutes earlier on reverse-phase HPLC and shows a +16 Da shift on mass spec. Many GHRH analogues have at least one Met; oxidation is a real storage-stability concern, not just a synthesis artifact.
- Deamidation. Asparagine and glutamine residues can deamidate to aspartate / iso-aspartate or glutamate over time, especially at neutral or basic pH. The peptide is intact in length but the side chain has changed and receptor activity may be altered. Common in older lots stored reconstituted.
- Racemization. Amino acid residues epimerizing from L to D form during coupling, especially at activated histidines or cysteines. The product looks correct by mass but is a stereoisomer with different binding affinity. Often indistinguishable from the parent without chiral analysis.
- TFA salts. Trifluoroacetic acid is the standard counterion in SPPS preparative HPLC. Most peptides ship as TFA salts unless explicitly converted to acetate. The TFA mass inflates the actual mass of the powder - a vial labelled "5 mg peptide" delivered as a TFA salt may contain 4.0–4.5 mg of free peptide and the rest as bound TFA. This affects your real dose; ask vendors for the salt form or for a free-base correction factor.
- Residual solvents. DMF, NMP, DCM, piperidine, and acetonitrile are SPPS workhorses. Trace amounts persist through lyophilisation. ICH Q3C limits exist for pharmaceutical- grade material; research-grade peptides are not held to those limits and may carry residual solvent loads in the hundreds of parts per million.
- Residual protecting groups. Side-chain protecting groups (Trt, Boc, Pbf) sometimes survive the global deprotection step. The peptide has an extra mass equal to the protecting group; you'll see the +M on mass spec if the lab looks for it, but standard "main peak" reporting may not flag it.
What the standard COA doesn't measure
HPLC + mass spec is the minimum acceptable test set for identity and bulk purity. It's not the complete safety set for an injectable. These three contaminant classes need separate tests that most research-grade COAs don't include:
- Endotoxin. Lipopolysaccharide from gram-negative bacterial contamination. Heat-stable, lyophilisation doesn't remove it. Tested by LAL (limulus amoebocyte lysate) or recombinant Factor C assay. Pharmacopoeial limits are typically expressed in EU/mg, with injectable thresholds around 5 EU/kg body weight per hour. Below that limit, endotoxin produces no notable response. Above it, fevers, malaise, and inflammation that mimic flu - sometimes mistaken for "TB-flu" or peptide side effects.
- Bioburden / sterility. Live bacterial or fungal contamination. Different from endotoxin (which is the membrane debris of dead bacteria); bioburden tests culture for growth. Lyophilised peptide rarely fails this test; reconstituted vials that pierce-cycle through dirty septa will.
- Heavy metals. Catalysts (Pd from coupling reagents) and contamination from raw amino acids. Tested by ICP-MS. Pharmaceutical limits in single-digit ppm; research-grade often not measured at all.
For peptides you'll inject more than a handful of times, the endotoxin add-on at the testing lab is worth the $50 it costs. Heavy metals testing is rarely worth pursuing on a single vial but is worth asking the manufacturer about for any lot you're building a long protocol around.
Research grade vs GMP grade
- Research grade means the manufacturer follows consistent processes for identity and bulk purity but does not operate under formal pharmaceutical-grade quality systems. ICH Q7 (active pharmaceutical ingredient GMP) doesn't apply. Documentation, change control, and audit trails are at the manufacturer's discretion. This covers the vast majority of the grey-market catalog.
- GMP grade means the manufacturing facility operates under the same quality system that produces approved pharmaceuticals: validated processes, batch records, deviation handling, supply-chain controls. Cost-of-goods is roughly 10–50× research grade for the same molecule.
- The gap that matters most: GMP-grade material has a paper trail you can audit. Research-grade material has whatever the COA says, and beyond that, trust. Both can produce the same molecule at the same purity for the same person on the same day. The differential is reproducibility across lots.
Practical implications for the user
- Demand a COA that lists the salt form. "5 mg peptide" can mean 4.0 mg free base + 1 mg TFA, or 4.7 mg free base + 0.3 mg acetate, or genuinely 5 mg free base. Your actual dose math depends on which.
- Treat purity numbers on long peptides with extra skepticism. A grey-market 35-mer labelled 99% main peak is doing something the math says is hard. It's not impossible - it just requires real preparative-HPLC work to land. If the price doesn't reflect that work, it likely isn't there.
- Pay for endotoxin testing on injection-heavy protocols. $50 once per lot beats a flu-mimicking inflammatory week mid-cycle. The most-skipped useful test on grey-market peptides.
- Watch for oxidation in older lots. If a vial that worked at month 1 stops responding at month 4, oxidation of a Met or Trp residue is a real possibility - not just dose drift or sourcing change. Reconstituted vials oxidise faster than lyophilised powder.
- Don't conflate "high purity" with "right peptide." A high purity number on the wrong identity is worse than a moderate purity number on the right one. The mass-spec readout is where identity lives; the HPLC main peak is where purity lives. Both have to land.
Decision frame
- Buying a short peptide (≤15 residues) from a vendor with
a real COA?
→ 98% main peak is a normal floor; 99.0–99.5% is achievable routinely. Below 98% on a short peptide is a quality signal worth investigating before committing to a multi-vial order. - Buying a long peptide (25+ residues) from a vendor with
a real COA?
→ 95% main peak is the realistic floor; 97–98% is good; 99% is plausible only with multiple purification passes. Compare the price-per-mg against the cost reality of multiple preparative-HPLC steps. - Planning a long protocol (months, daily injection)?
→ Pay for endotoxin testing on top of standard HPLC + mass spec. The marginal cost is small relative to the duration of exposure. - Suspecting a specific lot is bunk?
→ Send a vial for HPLC + mass spec. If main peak and identity both check, the next failure mode is salt-form mis-math (powder contains less free peptide than label) or oxidation drift (older reconstituted vial). Bioassay is the third check; see Sourcing and Verification.
What stops people
- Reading the COA without checking the lot number. A vendor recycling a sample COA across multiple batches is a red flag bigger than any purity number. Lot mismatch makes the rest of the document marketing.
- Comparing peptide purity numbers across length. "This 35-mer is 99% pure but this 7-mer is 98% pure, so the 35-mer is cleaner" - wrong frame. The synthesis ceiling is different. Compare like to like.
- Treating "research grade" as a downgrade phrase. A reputable research-grade manufacturer can produce material indistinguishable from GMP at the bench for any single lot. The GMP premium buys cross-lot consistency and audit trail, not necessarily purer molecule on day one.
- Skipping mass spec. HPLC purity without mass-spec identity tells you a clean unknown is in the vial. Most competent labs run both as a package; if a COA shows only one, ask why.
Cross-references
- Sourcing and Verification - the operator-level guide to actually running these tests and reading the COAs that come back.
- Cold Chain Reality - why oxidation and deamidation accelerate when storage rules slip, and how the impurity catalogue above grows over time in a poorly-stored vial.
- Reading a Trial Paper - companion literacy article on a different document type. Both teach the same skill: don't take a number at face value.
- Peptides 101 - the entry-point article if any of the synthesis vocabulary above is new ground.
What this article doesn't cover
Solution-phase synthesis (used for the smallest peptides and some cyclic ones) follows different efficiency math and isn't covered here. Recombinant production - the route most GMP semaglutide, tirzepatide, and somatropin take - has its own quality considerations (host cell protein contamination, glycosylation variants, viral-clearance validation) that don't apply to SPPS material. Specific vendor recommendations and current community testing-lab options are intentionally out of scope; both rotate faster than article publication can keep up with.